Iron Absorption
Iron metabolism is a tightly regulated biological system designed to balance two competing needs: the essential requirement of iron for oxygen transport and cellular respiration, and the potential toxicity of free iron when present in excess. Because the human body has no active excretory pathway for iron, control is exercised primarily at the level of intestinal absorption.
1. Iron Absorption in the Gastrointestinal Tract
Dietary iron absorption is inherently inefficient. On average, only about 10% of ingested iron is absorbed under normal physiological conditions. This efficiency varies significantly depending on dietary source:- Heme iron (animal sources) is absorbed more efficiently.
- Non-heme iron (plant sources) has lower and more variable absorption.
- Overall, iron from animal foods is absorbed at approximately twice the rate of iron from plant foods.
Chemical Forms of Dietary Iron
In food, iron is predominantly present in the ferric (Fe³⁺) state, often bound within:- Ferric hydroxides
- Organic iron complexes
However, ferric iron is poorly soluble and not readily absorbed. Therefore, the initial stages of digestion aim to convert iron into a more absorbable form.
2. Role of Gastric Environment and Reduction Reactions
In the stomach, the acidic environment created by gastric hydrochloric acid (HCl) and dietary organic acids helps liberate iron from food complexes. This process releases free ferric ions or loosely bound iron.
A crucial step follows: chemical reduction of Fe³⁺ to Fe²⁺ (ferrous iron).
This conversion is facilitated by reducing agents such as:
- Glutathione
- Other dietary and mucosal reducing compounds
Why Fe²⁺ Matters
Ferrous iron (Fe²⁺) is:
- More soluble in the intestinal environment
- More easily transported across the intestinal mucosa
- Therefore, the physiologically preferred absorptive form
Although gastric acid enhances iron availability, it is not absolutely essential. Clinical observations show that even in achlorhydria (absence of gastric acid), iron absorption may remain adequate, indicating compensatory mechanisms in the intestine.
3. Intestinal Uptake and the Concept of the Mucosal Block
Apoferritin and Ferritin Formation
Within intestinal mucosal cells, iron absorption was historically explained by the mucosal block hypothesis, centered on a protein called apoferritin.
Apoferritin is a large intracellular protein composed of approximately 20 peptide chains, with a molecular weight of about 460,000 Daltons.
It binds iron through the reaction:
Apoferritin + Fe³⁺ → Ferritin
This binding is reversible, allowing controlled storage and release of iron.
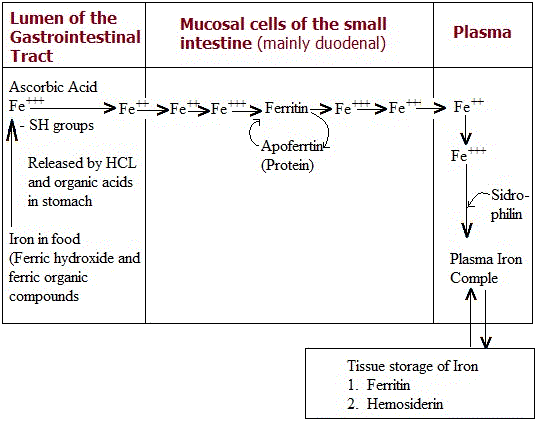
Ferritin as a Storage Form
Ferritin is a soluble iron-storage protein complex containing iron in the form of a ferric hydroxide–phosphate core. It can store iron efficiently, with iron constituting up to ~23% of its total weight.
Ferritin is widely distributed, particularly in:
- Intestinal mucosal cells
- Liver
- Spleen
- Bone marrow
It functions as a readily mobilizable iron reserve.
4. The Classical “Mucosal Block” Theory
According to the earlier model of regulation:- Iron enters mucosal cells and binds apoferritin to form ferritin.
- As apoferritin becomes saturated, further absorption slows.
- When ferritin breaks down, apoferritin is regenerated, continuing the cycle.
This led to the concept that iron absorption was limited by the availability of apoferritin, forming a “mucosal block” that prevented excessive iron entry into the body.
While this model provided an important early framework, it is now considered incomplete.
5. Modern Regulation: The Role of Hepcidin
Contemporary research has identified hepcidin, a peptide hormone produced by the liver, as the central regulator of systemic iron balance.
Hepcidin controls iron movement by acting on ferroportin, the only known cellular iron exporter located on:
- Enterocytes (intestinal cells)
- Macrophages (iron recycling cells)
When hepcidin levels are high:
- Ferroportin is degraded
- Iron export into plasma decreases
- Absorption from the intestine is reduced
When hepcidin levels are low:
- Ferroportin remains active
- Iron absorption and release increase
Thus, the hepcidin–ferroportin axis, rather than the mucosal block, is now understood to be the primary physiological control system.
The mucosal block theory remains valuable historically and conceptually but is no longer considered the main regulatory mechanism.
6. Exit from Enterocytes and Plasma Transport
Before iron leaves intestinal cells, it is maintained in the ferrous (Fe²⁺) state. Upon entry into the bloodstream, it undergoes oxidation back to ferric (Fe³⁺).
Role of Transferrin
In plasma, iron binds to a transport protein called transferrin (a β-globulin), with a molecular weight of approximately 90,000 Daltons.
Key properties of transferrin:
- Each molecule binds two iron atoms
- It serves as the primary iron transport system in circulation
- It delivers iron to tissues such as bone marrow for hemoglobin synthesis
Role of Ceruloplasmin
The oxidation of Fe²⁺ to Fe³⁺ is facilitated by ceruloplasmin, a copper-containing ferroxidase enzyme. This step is essential because transferrin binds iron only in the ferric state.
7. Iron Storage in the Body
Once transported, iron is either utilized or stored. The major storage forms include:
Ferritin
- Soluble and readily mobilizable
- Present in liver, spleen, bone marrow, and reticuloendothelial cells
- Represents the primary storage pool under normal conditions
Hemosiderin
When iron stores become excessive:
- Iron accumulates as hemosiderin
- It contains a higher iron concentration (~55%)
- It is insoluble and appears as coarse intracellular granules
- It is derived from aggregated ferritin degradation products
Hemosiderin deposits can be detected histologically using the Prussian blue reaction, which stains iron-containing compounds.
Summary
Iron absorption is a multi-step, highly regulated process involving:
- Conversion of dietary iron into absorbable ferrous form
- Uptake by intestinal mucosal cells
- Intracellular handling via ferritin
- Export regulated primarily by hepcidin
- Plasma transport via transferrin
- Storage in ferritin and hemosiderin complexes
Modern physiology emphasizes systemic hormonal control (hepcidin), while classical concepts like the mucosal block remain useful for foundational understanding of iron handling dynamics.




