Vitamin A – Occurrence and Absorption
Vitamin A is not a single chemical entity but rather a group of structurally related compounds that share similar biological activity. It is a fat-soluble alcohol, and in modern biochemical terminology the principal active form is referred to as retinol. Its oxidized aldehyde derivative is known as retinal, also called retinaldehyde.
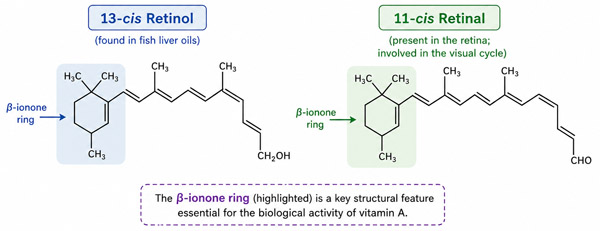
Structurally, vitamin A contains a characteristic ring system known as the β-ionone ring, which forms an essential part of its molecular framework and is crucial for its biological function, particularly in vision.
Vitamin A exists in multiple geometric forms (isomers), among which the most biologically and physiologically significant are:
- 13-cis retinol, which is commonly found in various fish liver oils
- 11-cis retinal, which is specifically present in the retina and plays a direct role in the visual cycle
From a chemical stability perspective, vitamin A is relatively heat-stable under normal cooking conditions. However, it becomes susceptible to destruction at high temperatures in the presence of oxygen (O₂) or air, where oxidative degradation significantly reduces its activity.
Vitamin A can be synthesized industrially, and in biological systems it is stored predominantly in the liver. In storage form, it exists mainly as retinyl esters, bound to cellular proteins, which allows safe sequestration without toxicity while maintaining a reserve supply for physiological needs.
1. Occurrence of Vitamin A
Vitamin A, in its preformed active form, is found naturally only in animal sources. It is particularly abundant in certain fish liver oils, where it occurs in very high concentrations. The richest dietary source is halibut liver oil, followed by other significant sources such as shark liver oil and cod liver oil. It is also present in the livers of other animals, as well as in egg yolk and milk, where it is primarily stored as retinyl esters.
In contrast, plants do not contain vitamin A in its active form. Instead, they provide precursor compounds known as provitamin A carotenoids (carotenes). These are yellow to red pigments widely distributed in plant-based foods such as:
- carrots
- yellow corn
- sweet potatoes
- peaches
- spinach
Although carotenes themselves do not possess direct vitamin A activity, they are biologically important because they are converted into vitamin A within the intestinal mucosa and liver after ingestion.
The efficiency of this conversion process can be influenced by metabolic conditions. It has classically been observed that conversion of carotenes to vitamin A is reduced in conditions such as diabetes mellitus and hypothyroidism, although more recent evidence suggests that the efficiency may vary considerably depending on overall nutritional status, metabolic balance, and individual physiology.
Among the different carotenoids, β-carotene is the most important and efficient precursor of vitamin A. This is due to its symmetrical molecular structure, in which each half of the molecule can yield one molecule of vitamin A upon enzymatic cleavage.
Despite this, carotenoids are considered relatively inefficient dietary sources of vitamin A because:
- their absorption in the intestine is incomplete
- their conversion to active vitamin A is not fully efficient
When consumed in excess, carotenoids may accumulate in adipose tissue and skin, leading to a harmless condition known as carotenemia, characterized by a yellowish discoloration of the skin without toxicity.
2. Absorption of Vitamin A
In natural dietary sources, vitamin A is commonly present in the form of retinyl esters. During digestion, these esters are hydrolyzed by esterases found in both pancreatic juice and the intestinal mucosa, resulting in the release of fatty acids and free vitamin A alcohol (retinol).
Absorption occurs primarily in the small intestine, where vitamin A is incorporated into micelles with the help of bile salts. This micellar incorporation is essential for solubilizing fat-soluble vitamins and enabling their uptake by intestinal cells.
Once inside the intestinal mucosal cells (enterocytes), vitamin A is re-esterified, reforming retinyl esters. These are then incorporated into chylomicrons, which enter the lymphatic system rather than directly entering the portal blood circulation. From the lymph, they are eventually delivered into the bloodstream.
In the circulation, vitamin A is transported mainly as retinol bound to retinol-binding protein (RBP). This binding is essential, as it:
- keeps retinol soluble in plasma
- protects it from degradation
- ensures targeted delivery to peripheral tissues
Carotenes follow a slightly different pathway. They are absorbed in the small intestine and then converted into retinal and retinol within both the intestinal mucosa and the liver.
The presence of bile salts is critical for efficient absorption of both vitamin A and carotenoids, as they facilitate micelle formation, which enhances lipid solubility and uptake.
The enzymatic conversion of β-carotene into vitamin A is catalyzed by β-carotene 15,15'-dioxygenase, an enzyme located in the intestinal mucosal cells, which cleaves the carotene molecule at its central double bond to produce retinal, which is then further reduced to retinol or oxidized depending on metabolic needs.
3. Metabolism of Vitamin A
Vitamin A metabolism refers to the sequence of biochemical transformations involving absorption, storage, interconversion, transport, utilization, and degradation of retinoids in the body. It is a tightly regulated process because both deficiency and excess of vitamin A can lead to significant clinical disorders.
1. Hepatic Uptake and Storage
After absorption, vitamin A is transported to the liver mainly in the form of chylomicron remnants. Hepatocytes and specialized liver cells called hepatic stellate cells (Ito cells) play a central role in its metabolism.
Inside the liver:
- Retinol is esterified with long-chain fatty acids to form retinyl esters
- These esters are stored intracellularly in lipid droplets of stellate cells
- The liver acts as the major storage organ, containing about 70–90% of total body vitamin A reserves
This storage form is biologically inactive and serves as a safe reserve that prevents toxicity.
2. Mobilization from the Liver
When peripheral tissues require vitamin A, it is mobilized from hepatic stores in a controlled manner:
- Retinyl esters are hydrolyzed back to retinol
- Retinol binds to Retinol-Binding Protein (RBP) within hepatocytes
- The retinol–RBP complex is then secreted into the bloodstream
In circulation, this complex further associates with transthyretin (prealbumin), which:
- stabilizes the complex
- prevents renal filtration and loss
- ensures efficient delivery to target tissues
This regulated transport prevents free retinol toxicity in plasma.
3. Peripheral Tissue Uptake and Intracellular Metabolism
Once delivered to target tissues, retinol enters cells via receptor-mediated uptake and undergoes one of several metabolic fates depending on tissue requirements.
(a) Conversion to Retinal (Visual Function)
In visual tissues, especially the retina:
- Retinol is oxidized to retinal (retinaldehyde)
- The 11-cis retinal is the biologically active form involved in the visual cycle
- It binds to opsin proteins to form rhodopsin, essential for low-light vision
This interconversion is reversible and highly regulated.
(b) Conversion to Retinoic Acid (Gene Regulation)
Retinol can also be irreversibly oxidized to retinoic acid, which acts as a hormone-like regulator.
Key features of retinoic acid:
- It is the active transcriptional form of vitamin A
- It binds to nuclear receptors:
- RAR (Retinoic Acid Receptors)
- RXR (Retinoid X Receptors)
- These receptors regulate gene expression involved in:
- epithelial cell differentiation
- embryonic development
- immune function
- growth regulation
Importantly:
- Retinoic acid cannot be converted back to retinol
- Therefore, it represents the end-point of vitamin A utilization pathway
(c) Re-esterification for Storage (Peripheral Tissues)
Some tissues can temporarily store vitamin A:
- Retinol may be re-esterified into retinyl esters
- However, the liver remains the principal storage site
4. Catabolism and Excretion
Vitamin A is a fat-soluble vitamin and is not easily excreted unchanged. Excess vitamin A undergoes slow metabolic breakdown:- Retinoic acid is oxidized to polar metabolites
- These metabolites are conjugated in the liver (glucuronidation)
- Excretion occurs mainly via:
- bile into feces (major route)
- small amounts in urine
Because elimination is slow, chronic excess intake can lead to hypervitaminosis A.
5. Key Regulatory Features of Vitamin A Metabolism
Vitamin A metabolism is tightly controlled at multiple levels:
- Hepatic release of retinol is RBP-dependent, preventing uncontrolled circulation
- Enzymatic conversion to retinal/retinoic acid is tissue-specific
- Retinoic acid regulates its own synthesis via feedback inhibition
- Storage in stellate cells prevents toxicity during high intake periods
Clinical Correlation
- Vitamin A deficiency → night blindness, xerophthalmia, keratinization of epithelium
- Excess vitamin A (hypervitaminosis A) → hepatotoxicity, bone pain, teratogenic effects, pseudotumor cerebri
- Protein deficiency (low RBP synthesis) → functional vitamin A deficiency despite adequate stores